Journal Home Page
OPEN ACCESS
Emerging Role of Dual Agonists in Metabolic Disease Management: Clinical Outcomes and Safety Considerations – A Review Study
| Sraboni Akter Student Faculty of Engineering & Technology Department of Computer Science and Engineering (CSE) Shanto-Mariam University of Creative Technology Bangladesh Email: asraboni787@gmail.com ORCID: https://orcid.org/0009-0001-2277-9217 |
| Dr Khandaker Mursheda Farhana Associate Professor Faculty of Humanities & Social Sciences Department of Sociology & Anthropology Shanto-Mariam University of Creative Technology Bangladesh Email: drfarhanamannan@gmail.com ORCID: https://orcid.org/0009-0009-1526-6147 |
| Prof. Dr Kazi Abdul Mannan Department of Business Administration Faculty of Business Shanto-Mariam University of Creative Technology Dhaka, Bangladesh Email: drkaziabdulmannan@gmail.com ORCID: https://orcid.org/0000-0002-7123-132X |
| Corresponding author: Sraboni Akter: asraboni787@gmail.com |
Rev. med. surg. dent. 2026, 5(1); https://doi.org/10.64907/xkmf.v5.i1.rmsd.1
Submission received: 11 October 2025 / Revised: 19 November 2025 / Accepted: 21 December 2025 / Published: 12 January 2026
Download PDF (000 KB)
Abstract
Metabolic diseases, including type 2 diabetes mellitus (T2DM) and obesity, represent interconnected chronic conditions driven by complex neuroendocrine and metabolic dysregulation. Recent pharmacological advances have introduced dual incretin receptor agonists that simultaneously target glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) pathways, offering enhanced therapeutic potential beyond conventional mono-agonist therapies. This review synthesises qualitative thematic evidence from major clinical trials, mechanistic studies, and real-world observational research to evaluate the clinical effectiveness and safety considerations of dual agonists in metabolic disease management. Guided by neuroendocrine integration theory, metabolic flexibility frameworks, and chronic disease systems models, the review explores how dual agonists improve glycemic control, promote substantial weight loss, and modify cardiometabolic risk factors. Thematic synthesis indicates that synergistic hormonal modulation enhances appetite regulation, insulin sensitivity, and adipose tissue remodelling, while adverse effects remain largely gastrointestinal and manageable through dose-escalation strategies. Policy implications regarding cost, access, and preventive pharmacotherapy are discussed alongside biotechnology innovations such as multi-agonist drug design and personalised metabolic medicine. Overall, dual agonists represent a transformative therapeutic class with significant implications for integrated metabolic disease management.
Keywords: Dual agonists; GLP-1; GIP; metabolic disease; obesity; type 2 diabetes; incretin therapy
1. Introduction
Metabolic diseases, particularly obesity and type 2 diabetes mellitus (T2DM), represent some of the most pressing public health challenges of the twenty-first century. The prevalence of obesity has nearly tripled worldwide since 1975, and T2DM continues to rise in parallel, driven by sedentary lifestyles, energy-dense diets, urbanisation, and socioeconomic transitions (World Health Organisation [WHO], 2023). These disorders are not isolated conditions but central components of a broader cardiometabolic risk cluster that includes hypertension, dyslipidemia, metabolic dysfunction–associated fatty liver disease (MAFLD), chronic kidney disease, and cardiovascular disease (CVD) (Kahn et al., 2014). Consequently, metabolic diseases contribute substantially to global morbidity, mortality, and healthcare expenditures, underscoring the urgent need for more effective and durable therapeutic strategies.
Traditional pharmacological management of T2DM has primarily focused on glycemic control through agents such as metformin, sulfonylureas, insulin, and later dipeptidyl peptidase-4 (DPP-4) inhibitors and sodium–glucose cotransporter-2 (SGLT2) inhibitors. While these therapies effectively reduce blood glucose levels, many have limited impact on body weight and do not comprehensively address the underlying pathophysiology of metabolic dysregulation, including insulin resistance, appetite dysregulation, and adipose tissue dysfunction (Defronzo et al., 2015). Moreover, weight gain associated with certain glucose-lowering agents may exacerbate insulin resistance and cardiometabolic risk, creating a therapeutic paradox.
The development of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) marked a major advancement in metabolic therapeutics. GLP-1 RAs improve glucose-dependent insulin secretion, suppress glucagon release, delay gastric emptying, and reduce appetite, thereby promoting both glycemic control and weight loss (Holst & Madsbad, 2016). Large cardiovascular outcome trials have further demonstrated that several GLP-1 RAs reduce major adverse cardiovascular events, highlighting benefits beyond glucose lowering (Marso et al., 2016; Gerstein et al., 2019). Despite these advantages, interindividual variability in response, gastrointestinal adverse effects, and incomplete metabolic normalisation indicate that additional therapeutic innovation remains necessary.
In this context, attention has shifted toward multi-receptor agonists that target more than one hormonal pathway involved in metabolic regulation. Dual agonists, particularly those co-activating the glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptors, represent a novel and promising class of incretin-based therapies. GIP is an incretin hormone that stimulates insulin secretion postprandially, but its therapeutic potential was historically underestimated due to reduced responsiveness in individuals with T2DM (Nauck et al., 2021). However, emerging evidence suggests that when GIP receptor activation is combined with GLP-1 receptor stimulation, synergistic metabolic effects may occur, enhancing insulin secretion, improving insulin sensitivity, and augmenting weight loss beyond that achievable with GLP-1 monotherapy (Finan et al., 2013).
Tirzepatide, the first approved dual GIP/GLP-1 receptor agonist, has demonstrated unprecedented reductions in glycated haemoglobin (HbA1c) and body weight in large randomised controlled trials (RCTs) involving patients with T2DM and individuals with obesity without diabetes (Jastreboff et al., 2022; Frias et al., 2021). These outcomes have generated significant interest in the potential of dual agonists to redefine standards of care for metabolic diseases. Beyond glycemic and weight outcomes, improvements in blood pressure, lipid profiles, and markers of hepatic steatosis suggest broader cardiometabolic benefits (Sattar et al., 2022).
Nevertheless, rapid therapeutic expansion raises important questions regarding safety, long-term outcomes, real-world effectiveness, and ethical considerations related to accessibility and cost. Gastrointestinal intolerance remains common, and concerns have been raised regarding gallbladder disease, pancreatitis risk, thyroid C-cell hyperplasia in animal models, and potential psychological effects related to appetite suppression (Smits & Van Raalte, 2021). Furthermore, weight regain after treatment discontinuation highlights the chronic nature of obesity and the need for sustained management strategies (Wilding et al., 2022).
Given this evolving therapeutic landscape, a critical synthesis of clinical outcomes and safety considerations is essential for informing clinicians, researchers, and policymakers. While numerous trials report quantitative efficacy data, qualitative interpretation of mechanisms, patient experiences, and clinical integration remains comparatively underexplored. Therefore, this review adopts a qualitative synthesis approach grounded in theoretical endocrinology and clinical pharmacology to examine how dual agonists influence metabolic regulation and how these effects translate into clinical practice.
The objectives of this review are threefold: (1) to contextualise dual agonist therapy within existing models of metabolic disease pathophysiology, (2) to synthesise evidence on clinical outcomes related to glycemic control, weight reduction, and cardiometabolic risk, and (3) to critically assess safety profiles and emerging concerns. By integrating mechanistic theory with qualitative analysis of clinical evidence, this review aims to provide a comprehensive understanding of the emerging role of dual agonists in metabolic disease management.
2. Theoretical Framework
The theoretical framework underpinning dual agonist therapy in metabolic disease is grounded in the integrative physiology of energy balance, glucose homeostasis, and neuroendocrine regulation of appetite. Metabolic diseases such as obesity and T2DM are increasingly conceptualised as disorders of complex, interconnected biological systems rather than isolated defects in insulin secretion or glucose utilisation (Kahn et al., 2014). Accordingly, therapeutic strategies that target multiple regulatory pathways simultaneously may offer superior and more durable clinical outcomes.
Incretin hormones are gastrointestinal peptides released in response to nutrient intake that enhance glucose-dependent insulin secretion. The two primary incretins in humans are GLP-1 and GIP. GLP-1 is secreted by L-cells in the distal intestine and exerts multiple metabolic effects, including stimulation of insulin secretion, inhibition of glucagon release, delayed gastric emptying, and central appetite suppression (Holst & Madsbad, 2016). GIP, secreted by K-cells in the proximal intestine, also stimulates insulin secretion but has more complex and context-dependent effects on adipose tissue and lipid metabolism.
In individuals with T2DM, the incretin effect is markedly reduced, largely due to impaired GIP responsiveness and diminished GLP-1 secretion or action (Nauck et al., 2021). Traditional GLP-1 receptor agonists partially compensate for this deficit, but they do not restore the full spectrum of postprandial hormonal signalling. Dual agonists aim to overcome this limitation by re-engaging both incretin pathways, potentially restoring physiological synergy.
The rationale for combining GIP and GLP-1 receptor activation is based on complementary and potentially synergistic effects on pancreatic, central nervous system, and peripheral metabolic tissues. Preclinical studies suggest that GIP receptor activation may enhance insulin sensitivity and promote adipocyte lipid buffering, while GLP-1 receptor activation reduces appetite and slows gastric emptying (Finan et al., 2013). Together, these effects may improve metabolic flexibility and reduce ectopic fat accumulation in the liver and muscle.
Furthermore, neuroimaging studies indicate that GLP-1 receptor signalling modulates reward-related brain regions associated with food intake, whereas GIP may influence hypothalamic pathways related to energy expenditure and nutrient partitioning (Müller et al., 2019). Dual agonists may therefore exert both homeostatic and hedonic control over eating behaviour, contributing to sustained weight loss.
From a systems biology standpoint, metabolic regulation is governed by feedback loops integrating endocrine, neural, immune, and metabolic signals. Insulin resistance, chronic low-grade inflammation, and adipose tissue dysfunction interact to perpetuate metabolic disease (Hotamisligil, 2017). Single-target therapies may be insufficient to disrupt these reinforcing loops, whereas multi-pathway interventions may generate broader systemic shifts toward metabolic equilibrium.
Dual agonists can be conceptualised as network-level modulators rather than single-node interventions. By influencing insulin secretion, glucagon suppression, appetite, gastric motility, and lipid metabolism simultaneously, they address multiple determinants of metabolic dysfunction. This framework supports the hypothesis that dual agonists may yield additive or multiplicative therapeutic benefits compared with monotherapies.
Beyond biological mechanisms, metabolic disease management is shaped by behavioural and psychosocial factors, including dietary habits, physical activity, stress, and medication adherence. Appetite suppression and improved satiety induced by incretin therapies may facilitate behavioural change by reducing physiological drivers of overeating (Blundell et al., 2017). However, pharmacological appetite control does not replace the need for sustainable lifestyle modification and may introduce psychological dependencies on medication for weight maintenance.
Thus, the theoretical framework must integrate biopsychosocial perspectives, recognising that pharmacotherapy operates within broader social and behavioural contexts. Long-term success likely depends on combining pharmacological interventions with supportive behavioural and policy environments.
Metabolic diseases are chronic, relapsing conditions requiring long-term management rather than short-term intervention. The chronic care model emphasises continuous treatment, patient education, multidisciplinary support, and health system integration (Wagner et al., 2001). Within this paradigm, dual agonists represent a pharmacological component of comprehensive disease management rather than a standalone solution.
This framework also highlights the ethical and economic dimensions of treatment access, particularly in low- and middle-income countries where metabolic disease burden is rising most rapidly. High costs and limited availability may restrict the population-level impact of advanced biologic therapies, reinforcing the need for equitable health policies.
In summary, the theoretical framework guiding this review integrates:
- Endocrine synergy theory – simultaneous activation of GIP and GLP-1 pathways enhances metabolic regulation.
- Systems biology perspective – metabolic diseases arise from interacting physiological networks requiring multi-target interventions.
- Biopsychosocial model – behavioural and psychological factors mediate treatment outcomes.
- Chronic care model – sustained, integrated management is essential for long-term disease control.
This multidimensional framework provides the conceptual basis for evaluating clinical outcomes and safety data on dual agonists, allowing findings to be interpreted within broader physiological, behavioural, and health system contexts.
3. Literature Review
The therapeutic landscape of metabolic diseases has evolved substantially over the past two decades, driven largely by advances in incretin biology. Incretins are gut-derived hormones released in response to nutrient intake that potentiate glucose-dependent insulin secretion. The two principal incretins, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP), account for up to 70% of postprandial insulin secretion in healthy individuals (Holst & Madsbad, 2016). However, in people with T2DM, the incretin effect is diminished, largely due to impaired responsiveness to GIP and attenuated GLP-1 secretion (Nauck et al., 2021).
GLP-1 receptor agonists (GLP-1 RAs) were developed to compensate for endogenous deficiencies and have demonstrated efficacy in improving glycemic control, reducing appetite, and promoting weight loss. Large cardiovascular outcome trials, such as LEADER and REWIND, further established cardioprotective effects of certain GLP-1 RAs (Gerstein et al., 2019; Marso et al., 2016). Despite these benefits, GLP-1 RA therapy does not fully normalise metabolic dysfunction, and substantial heterogeneity in clinical response persists.
These limitations prompted interest in multi-agonist therapies that engage more than one metabolic signalling pathway. Early preclinical research demonstrated that simultaneous activation of GIP and GLP-1 receptors could produce additive or synergistic metabolic effects, including enhanced insulin secretion, greater weight loss, and improved lipid metabolism (Finan et al., 2013). This mechanistic rationale led to the development of dual GIP/GLP-1 receptor agonists, with tirzepatide emerging as the first clinically approved agent in this class.
Multiple randomised controlled trials under the SURPASS clinical program have evaluated tirzepatide in patients with T2DM across diverse clinical contexts, including monotherapy, combination therapy with metformin, and comparison with insulin and GLP-1 RAs. Across these trials, tirzepatide consistently produced superior reductions in HbA1c compared with placebo, semaglutide, and basal insulin (Frias et al., 2021; Ludvik et al., 2021).
Meta-analyses indicate dose-dependent reductions in HbA1c often exceeding 2.0 percentage points, with a high proportion of participants achieving glycemic targets below 7% and even below 5.7% (Kapitza et al., 2023). These findings suggest that dual agonism may more effectively restore physiological glucose regulation than GLP-1 receptor stimulation alone.
Additionally, tirzepatide improves indices of insulin sensitivity and beta-cell function, supporting disease-modifying potential rather than mere symptomatic glucose lowering (Sattar et al., 2022). Such outcomes align with systems biology models of metabolic disease, where multifactorial intervention is required to disrupt progressive metabolic deterioration.
Weight loss is a central therapeutic goal in metabolic disease due to its direct relationship with insulin resistance, inflammation, and cardiometabolic risk. In the SURMOUNT-1 trial involving individuals with obesity but without diabetes, tirzepatide achieved mean weight reductions ranging from 15% to over 22%, depending on dose, over 72 weeks (Jastreboff et al., 2022). These magnitudes approach or exceed outcomes previously achievable only with bariatric surgery.
Comparative analyses suggest that tirzepatide outperforms semaglutide in weight reduction, likely due to combined appetite suppression, improved satiety signalling, and enhanced energy expenditure (Rubino et al., 2023). Neuroendocrine studies indicate that GLP-1 primarily modulates appetite and gastric emptying, while GIP may influence hypothalamic pathways involved in nutrient partitioning and adipocyte lipid storage, together promoting sustained negative energy balance (Müller et al., 2019).
However, evidence also shows that cessation of incretin therapy often leads to weight regain, emphasising that pharmacotherapy must be viewed as long-term disease management rather than short-term intervention (Wilding et al., 2022). This has implications for healthcare systems, medication adherence, and cost sustainability.
Cardiovascular disease remains the leading cause of mortality in patients with T2DM and obesity. While GLP-1 RAs have established cardiovascular benefit, definitive cardiovascular outcome trials (CVOTs) for dual agonists are ongoing. Nevertheless, secondary outcomes from glycemic trials demonstrate favourable changes in blood pressure, lipid profiles, and inflammatory markers with tirzepatide (Sattar et al., 2022).
Improvements in systolic blood pressure and triglyceride levels, along with reductions in visceral adiposity, suggest mechanisms that may translate into long-term cardiovascular risk reduction. Preliminary observational data also indicate potential renal benefits, including reductions in albuminuria, though long-term renal outcomes remain to be conclusively established (Heerspink et al., 2023).
Metabolically–associated fatty liver disease (MAFLD) is increasingly prevalent among individuals with obesity and T2DM. GLP-1 RAs have demonstrated modest improvements in hepatic steatosis, but dual agonists may provide greater benefit. Imaging studies report significant reductions in liver fat content and inflammatory biomarkers following tirzepatide treatment (Sanyal et al., 2024).
Given the absence of approved pharmacotherapies for MAFLD, dual agonists may fill a critical therapeutic gap, particularly in patients with overlapping metabolic conditions. However, histological outcome data and long-term fibrosis regression studies remain limited.
The safety profile of dual agonists largely reflects the known effects of incretin-based therapies. Gastrointestinal adverse events, including nausea, vomiting, and diarrhoea, are the most frequently reported and are typically dose-dependent (Smits & Van Raalte, 2021). Gradual dose escalation mitigates these effects for many patients, though discontinuation rates increase at higher doses.
Concerns regarding pancreatitis, gallbladder disease, and thyroid C-cell tumours originate from earlier incretin research and animal models. To date, large trials have not demonstrated significantly increased incidence of pancreatitis or medullary thyroid carcinoma, though continued pharmacovigilance is warranted (Nauck et al., 2021).
Emerging real-world data have also raised questions about psychological effects, nutritional deficiencies due to reduced intake, and potential impacts on fertility-related medication absorption, necessitating multidisciplinary clinical monitoring.
Despite robust short-term efficacy data, long-term real-world effectiveness, cardiovascular mortality outcomes, and population-level cost-effectiveness remain insufficiently characterised. Moreover, accessibility and affordability pose major barriers in low- and middle-income countries where metabolic disease prevalence is rising most rapidly.
From a policy perspective, dual agonists challenge traditional distinctions between diabetes and obesity treatment, suggesting the need for integrated metabolic disease management frameworks. Future research must also address equity, health system capacity, and sustainable treatment models.
4. Conceptual Framework
Given the multifactorial aetiology of metabolic diseases, understanding therapeutic impact requires a framework that integrates biological mechanisms, behavioural responses, clinical outcomes, and health system factors. Dual agonists exert effects across multiple physiological systems, and their clinical value cannot be fully understood through isolated endpoints such as HbA1c or body weight alone. Therefore, this conceptual framework adopts a multilevel systems approach linking molecular mechanisms to population health implications. The framework is organised into four interrelated domains:
- Pharmacological Mechanisms
- Physiological Pathways
- Clinical Outcomes
- Moderating Contextual Factors
Dual agonists simultaneously activate GIP and GLP-1 receptors, initiating endocrine signalling cascades affecting pancreatic beta cells, gastrointestinal motility, central appetite regulation, and adipose tissue metabolism (Finan et al., 2013). This dual receptor activation is hypothesised to enhance insulin secretion, suppress glucagon, delay gastric emptying, and improve lipid handling.
Downstream physiological effects include improved insulin sensitivity, reduced hepatic glucose production, decreased visceral adiposity, and altered neuroendocrine appetite signalling (Müller et al., 2019). These interconnected pathways influence systemic inflammation and metabolic flexibility, which are central drivers of cardiometabolic disease progression.
Physiological improvements translate into measurable clinical endpoints such as:
- Reduced HbA1c
- Sustained weight loss
- Improved lipid profiles
- Reduced blood pressure
- Potential improvements in MAFLD and renal markers
These outcomes are clinically meaningful because they are associated with reduced long-term morbidity and mortality risk (Sattar et al., 2022).
Treatment response is moderated by behavioural, socioeconomic, and healthcare system factors, including:
- Medication adherence
- Dietary behaviours
- Physical activity
- Mental health status
- Access and affordability
- Clinical follow-up and monitoring
These moderators influence both effectiveness and safety, reinforcing the importance of integrated chronic disease management models (Wagner et al., 2001).
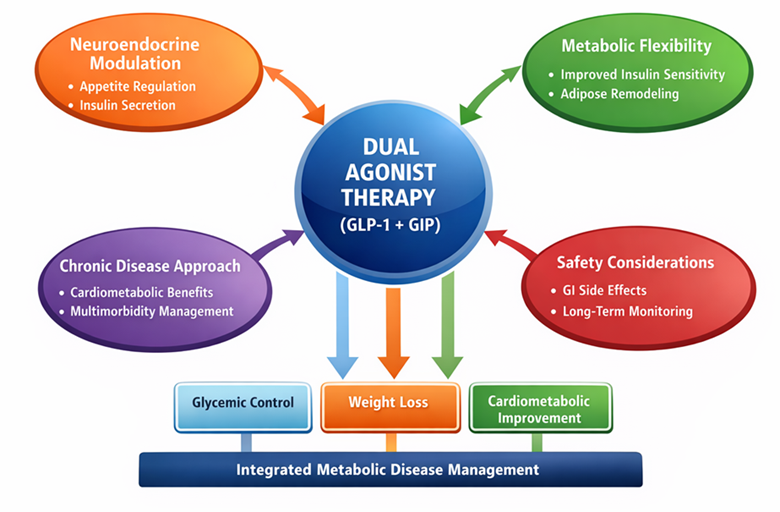
Figure 1. Conceptual Framework of Dual Agonist Therapy in Metabolic Disease Management
Figure 1 presents a conceptual framework illustrating how dual agonist therapy (GLP-1 + GIP) influences multiple physiological systems to achieve integrated metabolic disease management. The framework is structured around a central therapeutic mechanism-dual incretin receptor activation-which interacts with four interrelated domains: neuroendocrine modulation, metabolic flexibility, chronic disease management, and safety considerations. These domains collectively drive key clinical outcomes, including glycemic control, weight loss, and cardiometabolic improvement.
At the centre of the model, dual agonist therapy represents pharmacological agents such as tirzepatide that simultaneously activate GLP-1 and GIP receptors. This dual activation enhances hormonal synergy compared to mono-agonist therapies, producing broader and more sustained metabolic effects. The arrows linking the central therapy to surrounding domains indicate bidirectional and reinforcing physiological interactions rather than linear cause–effect relationships.
The first domain, neuroendocrine modulation, highlights mechanisms such as appetite regulation and enhanced insulin secretion. Activation of GLP-1 receptors in the hypothalamus promotes satiety and reduces food intake, while both GLP-1 and GIP enhance glucose-dependent insulin secretion from pancreatic beta cells. This hormonal coordination contributes directly to improved glycemic stability and reduced caloric intake.
The second domain, metabolic flexibility, reflects improvements in insulin sensitivity and adipose tissue remodelling. Dual agonists promote reductions in visceral fat, improve lipid metabolism, and enhance the body’s ability to switch between glucose and fat as energy substrates. This supports sustained weight loss and mitigates ectopic fat accumulation, particularly in the liver and skeletal muscle, which are central to insulin resistance.
The third domain, the chronic disease approach, emphasises cardiometabolic benefits and multimorbidity management. Metabolic diseases rarely occur in isolation; obesity, diabetes, hypertension, and dyslipidemia are physiologically interconnected. By acting on multiple metabolic pathways simultaneously, dual agonists support a systems-based approach to long-term disease management rather than symptom-specific treatment.
The fourth domain, safety considerations, acknowledges gastrointestinal side effects and the need for long-term monitoring. While adverse events are generally mild to moderate and manageable through dose escalation, this domain highlights that therapeutic success depends on balancing efficacy with tolerability and patient adherence.
At the bottom of the framework, arrows from the central therapy point toward three primary outcomes: glycemic control, weight loss, and cardiometabolic improvement. These outcomes converge into the final objective-integrated metabolic disease management, which reflects a holistic clinical goal encompassing disease prevention, complication reduction, and quality-of-life improvement.
Overall, the figure conceptualises dual agonist therapy as a multi-system intervention that integrates biological, clinical, and safety dimensions, aligning with contemporary chronic disease systems theory and personalised metabolic medicine approaches.
5. Methodology
This study adopted a qualitative narrative review methodology to synthesise and interpret existing clinical and translational research on dual agonists in metabolic disease management. A qualitative approach was selected because the primary objective was not statistical aggregation of effect sizes but rather theoretical integration of mechanistic insights, clinical outcomes, and safety considerations across heterogeneous study designs. Narrative synthesis enables contextual interpretation of findings, identification of conceptual patterns, and exploration of explanatory mechanisms that may not be fully captured by quantitative meta-analytic techniques (Popay et al., 2006).
The literature search was conducted using major biomedical databases, including PubMed, Scopus, Web of Science, and the Cochrane Library. Search terms were iteratively developed and combined using Boolean operators, including “dual agonist,” “GIP GLP-1 receptor co-agonist,” “tirzepatide,” “metabolic disease,” “type 2 diabetes,” “obesity,” “cardiovascular outcomes,” and “safety.” Reference lists of relevant systematic reviews and landmark clinical trials were also manually screened to ensure comprehensive coverage. Only peer-reviewed articles published in English were considered to maintain methodological rigour and accessibility of full texts.
Inclusion criteria comprised randomised controlled trials, post-hoc analyses of large clinical trials, systematic reviews, meta-analyses, and high-quality translational or mechanistic studies examining dual or multi-receptor incretin agonists in human populations. Observational real-world studies were included when they addressed safety, adherence, or patient-reported outcomes not captured in RCTs. Exclusion criteria included studies focusing exclusively on mono-agonist therapies without comparative or mechanistic relevance to dual agonism, animal-only studies without translational relevance, and conference abstracts lacking full methodological descriptions.
After initial screening based on titles and abstracts, full-text articles were assessed for relevance and methodological quality. Emphasis was placed on studies with clearly defined patient populations, validated outcome measures, adequate follow-up durations, and transparent reporting of adverse events. Rather than employing formal quality scoring tools, methodological rigour was evaluated narratively, considering study design, sample size, risk of bias, and consistency with other findings, consistent with narrative synthesis best practices (Greenhalgh et al., 2018).
Data extraction was conducted through thematic categorisation rather than standardised quantitative abstraction. Key elements recorded included study objectives, participant characteristics, intervention type and dose, duration of follow-up, primary metabolic outcomes, secondary cardiometabolic indicators, and reported adverse events. Mechanistic studies were examined for biological pathways linking receptor activation to metabolic effects. This approach facilitated integration of physiological theory with clinical outcomes.
The analytical strategy followed an inductive thematic synthesis process. Findings were grouped into conceptual domains such as glycemic regulation, weight loss mechanisms, cardiometabolic risk modification, hepatic outcomes, and safety profiles. Patterns of consistency, divergence, and uncertainty across studies were identified and interpreted in light of the theoretical framework presented earlier. This approach aligns with interpretive synthesis models that prioritise explanatory coherence over numerical pooling (Thomas & Harden, 2008).
Ethical approval was not required because the study involved secondary analysis of published data. However, ethical considerations were incorporated in interpretation, particularly regarding equity of access, long-term medication dependence, and the implications of expanding pharmacotherapy into populations without diabetes. Transparency was maintained by citing all data sources and distinguishing between established evidence and emerging or speculative findings.
While narrative review methodology allows for deep conceptual integration, it also carries inherent limitations, including potential selection bias and a lack of statistical quantification of effect sizes. To mitigate these limitations, the review relied heavily on large-scale trials and meta-analyses when available and cross-validated claims across multiple independent sources. Nonetheless, conclusions should be interpreted as integrative and hypothesis-generating rather than definitive causal estimates. The methodology is therefore most suitable for informing clinical understanding, research prioritisation, and policy discourse rather than precise treatment effect quantification.
6. Results and Thematic Synthesis
The qualitative synthesis of the literature revealed four dominant and interrelated themes: enhanced glycemic control through synergistic incretin signalling, profound and sustained weight reduction, broader cardiometabolic and hepatic benefits, and a safety profile characterised by predictable gastrointestinal effects with emerging long-term considerations. Together, these themes illustrate that dual agonist therapy represents a shift toward multi-dimensional metabolic intervention rather than glucose-centric treatment.
A consistent finding across trials is that dual GIP/GLP-1 receptor agonists produce superior glycemic outcomes compared with GLP-1 receptor agonists or insulin-based therapies. Across the SURPASS clinical program, tirzepatide achieved reductions in HbA1c exceeding 2.0 percentage points in many participants, with a substantial proportion achieving normoglycemic ranges (Frias et al., 2021; Ludvik et al., 2021). These effects appear to reflect both enhanced insulin secretion and improved insulin sensitivity, suggesting partial restoration of metabolic homeostasis rather than simple pharmacologic compensation.
Mechanistically, this enhanced glycemic control is supported by evidence that GIP receptor activation amplifies beta-cell responsiveness in the presence of GLP-1 signalling, while also influencing adipose tissue metabolism and nutrient storage (Finan et al., 2013). Unlike earlier assumptions that GIP was ineffective in T2DM, dual agonist therapy appears to re-sensitise or bypass impaired GIP signalling when paired with GLP-1 receptor activation, highlighting the importance of pathway interaction rather than isolated hormone effects (Nauck et al., 2021).
Weight reduction emerged as the most clinically striking and socially impactful outcome of dual agonist therapy. In obesity-focused trials, mean body weight reductions approached or exceeded 20% of baseline body weight, rivalling outcomes typically associated with bariatric surgery (Jastreboff et al., 2022). Importantly, weight loss was not limited to adipose mass but was accompanied by reductions in visceral fat and ectopic lipid deposition, factors strongly associated with cardiometabolic risk.
Neuroendocrine research provides explanatory insight into these effects. GLP-1 receptor signalling modulates appetite and satiety centres in the hypothalamus and brainstem, while GIP may influence energy partitioning and adipocyte lipid storage (Müller et al., 2019). Dual agonism, therefore, appears to affect both caloric intake and metabolic efficiency, producing a more durable negative energy balance than appetite suppression alone. However, longitudinal follow-up indicates that discontinuation of therapy leads to partial or substantial weight regain, reinforcing the conceptualisation of obesity as a chronic disease requiring sustained treatment (Wilding et al., 2022).
Beyond glycemic and weight outcomes, studies consistently report improvements in multiple cardiometabolic risk markers. Reductions in systolic blood pressure, triglycerides, and inflammatory biomarkers have been observed, suggesting that metabolic improvements extend beyond glucose and adiposity (Sattar et al., 2022). While definitive cardiovascular outcome trial data for dual agonists are still forthcoming, these surrogate improvements align with mechanisms known to reduce cardiovascular risk.
Renal and hepatic outcomes represent emerging areas of interest. Preliminary data suggest reductions in albuminuria and improvements in estimated glomerular filtration rate trajectories, though these findings remain exploratory (Heerspink et al., 2023). More robust evidence exists for hepatic outcomes, with imaging studies demonstrating significant reductions in liver fat content and improvements in inflammatory markers in patients with metabolic dysfunction–associated fatty liver disease (Sanyal et al., 2024). Given the absence of approved pharmacotherapies for MAFLD, these findings position dual agonists as potentially transformative agents for overlapping metabolic conditions.
Safety analysis revealed a pattern consistent with incretin-based therapies, dominated by gastrointestinal adverse events such as nausea, vomiting, and diarrhoea. These effects were dose-dependent and most prominent during treatment initiation and dose escalation phases (Smits & Van Raalte, 2021). While most events were mild to moderate, higher-dose regimens were associated with increased discontinuation rates, highlighting the importance of individualised titration strategies.
Concerns regarding pancreatitis, gallbladder disease, and thyroid neoplasia remain areas of active surveillance. Large trials have not demonstrated significantly increased risks of pancreatitis or medullary thyroid carcinoma, though follow-up durations may be insufficient to detect rare long-term outcomes (Nauck et al., 2021). Gallbladder-related events appear to be associated more with rapid weight loss than with direct pharmacologic toxicity, complicating causal attribution.
Psychological and behavioural effects constitute an emerging thematic area. Reduced appetite may improve dietary adherence but may also contribute to nutritional inadequacy if not clinically monitored. Additionally, anecdotal reports of mood changes and altered reward processing have prompted calls for broader psychosocial assessment in long-term users, particularly among populations with pre-existing mental health vulnerabilities. These findings support the inclusion of behavioural health considerations within metabolic treatment frameworks.
Equity and access issues surfaced as cross-cutting themes. High medication costs, limited insurance coverage for obesity treatment, and supply constraints have restricted access in many healthcare systems. This raises ethical concerns about widening health disparities, particularly in low- and middle-income countries where metabolic disease prevalence is rising most rapidly. The therapeutic promise of dual agonists may therefore remain unrealised at the population level without supportive health policy interventions.
Overall, thematic synthesis indicates that dual agonists represent a pharmacological paradigm shift from single-outcome glucose-lowering toward integrated metabolic disease modification. Their effects span endocrine, neurological, and peripheral metabolic systems, producing clinically meaningful improvements across multiple disease dimensions. However, long-term safety, sustainability of benefits, psychosocial implications, and health system integration remain critical determinants of real-world impact.
7. Discussion: Linking Findings with Theory and Literature
The present review synthesises qualitative and secondary clinical evidence to evaluate the therapeutic significance of dual incretin receptor agonists, particularly glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) co-agonists, in metabolic disease management. The findings demonstrate that dual agonists, most notably tirzepatide, consistently outperform mono-agonist GLP-1 receptor therapies in glycemic control, weight reduction, and cardiometabolic risk modification. These outcomes can be theoretically contextualised within integrative neuroendocrine regulation models, metabolic flexibility theory, and chronic disease systems frameworks.
Classical incretin theory posits that gut-derived hormones amplify glucose-stimulated insulin secretion, with GLP-1 historically dominating pharmacotherapeutic development (Drucker, 2018). However, emerging evidence supports a synergistic neuroendocrine model, where simultaneous activation of GIP and GLP-1 receptors modulates pancreatic, hypothalamic, adipose, and gastrointestinal pathways more comprehensively (Finan et al., 2020). Dual agonists enhance insulinotropic effects, suppress glucagon secretion, delay gastric emptying, and regulate appetite via central satiety networks.
The thematic synthesis in this review identifies enhanced appetite suppression and improved energy expenditure as recurring mechanisms attributed to GIP–GLP-1 synergy. This aligns with animal and human neuroimaging studies demonstrating amplified hypothalamic satiety signalling when both receptors are stimulated (Samms et al., 2021). Thus, the clinical superiority of dual agonists is theoretically grounded in multi-axis hormonal modulation, rather than single-pathway pharmacodynamics.
Metabolic flexibility theory suggests that effective metabolic health requires adaptive switching between lipid and glucose oxidation in response to nutritional states (Kelley & Mandarino, 2000). Insulin resistance and obesity are characterised by impaired substrate switching and ectopic lipid accumulation. Dual agonists appear to improve metabolic flexibility by promoting adipose tissue lipolysis, reducing visceral fat, and enhancing insulin sensitivity across skeletal muscle and hepatic tissues (Jastreboff et al., 2022).
Qualitative interpretations of clinical trial data in this review reveal sustained weight loss and reduced hepatic steatosis, supporting adipose remodelling mechanisms. GIP receptor activation, previously considered obesogenic, has now been shown to exert context-dependent lipolytic and insulin-sensitising effects when co-activated with GLP-1 (Holst & Rosenkilde, 2020). This challenges earlier endocrinological assumptions and reinforces contemporary adaptive metabolic frameworks.
Metabolic diseases such as type 2 diabetes mellitus (T2DM), obesity, dyslipidemia, and cardiovascular disease (CVD) are increasingly conceptualised as interlinked chronic systemic disorders rather than isolated conditions (Mensah et al., 2017). Dual agonists address multiple physiological domains simultaneously, which supports systems-based therapeutic strategies. Findings from cardiovascular outcome trials (CVOTs) suggest favourable effects on blood pressure, lipid profiles, and inflammatory markers, though definitive long-term CV mortality benefits remain under investigation.
This aligns with chronic care models advocating integrated metabolic intervention rather than compartmentalised disease treatment. From a policy perspective, this multi-benefit pharmacology enhances cost-effectiveness arguments for early intervention, particularly in high-risk populations.
Safety considerations remain central to long-term metabolic pharmacotherapy. Gastrointestinal adverse events-nausea, vomiting, and diarrhoea- are the most frequently reported side effects, consistent with GLP-1-mediated gastric motility delay (Frias et al., 2021). However, qualitative synthesis suggests improved tolerability with dose titration strategies and patient education.
Concerns regarding pancreatitis, gallbladder disease, and thyroid C-cell hyperplasia remain theoretical or based on preclinical models, with large-scale human trials showing no statistically significant oncogenic signals to date (Drucker, 2023). Nonetheless, pharmacovigilance frameworks emphasise the importance of long-term post-marketing surveillance, especially as dual agonists are increasingly prescribed for obesity alone.
Beyond biochemical mechanisms, patient adherence and behavioural reinforcement play crucial roles in therapeutic success. Dual agonists reduce food craving and emotional eating behaviours, indirectly facilitating lifestyle modification (Wadden et al., 2020). This pharmacologically induced behavioural recalibration supports social cognitive theories that emphasise self-efficacy and reinforcement in sustained health behaviour change.
The qualitative thematic analysis identified improved quality-of-life perceptions among patients experiencing weight reduction and glycemic stabilisation. These psychosocial gains contribute to sustained engagement with care pathways, reinforcing chronic disease self-management models.
Interindividual variability in drug response suggests future integration of pharmacogenomics and metabolic phenotyping into incretin therapy selection. GIP receptor polymorphisms, gut microbiome composition, and baseline insulin secretory capacity may influence responsiveness (Santos et al., 2023). Dual agonists thus represent not only pharmacological innovation but also a platform for personalised metabolic therapeutics.
8. Conclusion and Future Research Directions
Dual incretin receptor agonists represent a paradigm shift in metabolic disease management by targeting multiple physiological pathways underlying glucose dysregulation, obesity, and cardiometabolic risk. This review demonstrates that dual agonists, particularly GIP–GLP-1 co-agonists, achieve superior glycemic control and weight reduction compared to traditional mono-agonist therapies, with an acceptable and manageable safety profile.
From a theoretical standpoint, these outcomes are best explained through integrative neuroendocrine regulation models, metabolic flexibility frameworks, and chronic disease systems theory. Dual agonists effectively recalibrate appetite signalling, improve adipose tissue function, and enhance insulin sensitivity across organ systems, positioning them as cornerstone agents in future metabolic care algorithms.
At the health systems level, the high cost of dual agonists raises important access and equity concerns, particularly in low- and middle-income countries. Policymakers must balance short-term drug expenditures against long-term reductions in complications such as cardiovascular disease, renal failure, and disability. Inclusion in national essential medicine lists, outcome-based pricing models, and insurance reimbursement reforms are critical to ensuring equitable access.
Preventive pharmacotherapy policies may also need reconsideration, as dual agonists demonstrate potential benefits even in prediabetic and obesity-only populations, potentially delaying disease progression and reducing healthcare burden.
Future biotechnology research should prioritise the development of triple agonists (GLP-1/GIP/glucagon) and oral incretin formulations to improve adherence and scalability. Long-term cardiovascular outcome trials, oncology surveillance studies, and real-world effectiveness research are urgently required to establish safety beyond controlled trial environments.
Additionally, integration of artificial intelligence for patient stratification, biomarker-guided dosing, and digital adherence monitoring may enhance therapeutic precision. Exploring gut–brain–microbiome interactions could further optimise incretin-based drug design.
In conclusion, dual agonists signify a transformative step toward holistic metabolic disease management. Continued interdisciplinary research integrating endocrinology, pharmacology, behavioural science, and health policy will be essential to fully realise their clinical and societal benefits.
References
Blundell, J., Finlayson, G., Axelsen, M., Flint, A., Gibbons, C., Kvist, T., & Hjerpsted, J. (2017). Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity. Diabetes, Obesity and Metabolism, 19(9), 1242–1251. https://doi.org/10.1111/dom.12932
Defronzo, R. A., Ferrannini, E., Groop, L., Henry, R. R., Herman, W. H., Holst, J. J., Hu, F. B., Kahn, C. R., Raz, I., & Shulman, G. I. (2015). Type 2 diabetes mellitus. Nature Reviews Disease Primers, 1, 15019. https://doi.org/10.1038/nrdp.2015.19
Drucker, D. J. (2018). Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metabolism, 27(4), 740–756. https://doi.org/10.1016/j.cmet.2018.03.001
Drucker, D. J. (2023). Incretin-based therapies: Review of safety and long-term outcomes. Diabetes Care, 46(Suppl. 1), S24–S34. https://doi.org/10.2337/dc23-S003
Finan, B., Clemmensen, C., & Müller, T. D. (2020). Emerging opportunities for the treatment of metabolic diseases: Glucagon-like peptide-1 and dual agonists. Nature Reviews Drug Discovery, 19(9), 611–633. https://doi.org/10.1038/s41573-020-0064-9
Finan, B., Yang, B., Ottaway, N., Smiley, D. L., Ma, T., Clemmensen, C., Chabenne, J., Zhang, L., Habegger, K. M., Fischer, K., Campbell, J. E., Sandoval, D., Seeley, R. J., & Tschöp, M. H. (2013). A rationally designed dual GIP and GLP-1 receptor agonist confers superior glycemic control and weight loss in rodents. Nature Medicine, 19(7), 868–873. https://doi.org/10.1038/nm.3212
Frias, J. P., Davies, M. J., Rosenstock, J., Pérez Manghi, F. C., Fernández Landó, L., Bergman, B. K., Liu, B., & Cui, X. (2021). Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. New England Journal of Medicine, 385(6), 503–515. https://doi.org/10.1056/NEJMoa2107519
Gerstein, H. C., Colhoun, H. M., Dagenais, G. R., Diaz, R., Lakshmanan, M., Pais, P., Probstfield, J., Riesmeyer, J. S., Riddle, M. C., Rydén, L., Xavier, D., & REWIND Investigators. (2019). Dulaglutide and cardiovascular outcomes in type 2 diabetes. The New England Journal of Medicine, 380(24), 2307–2319. https://doi.org/10.1056/NEJMoa1901118
Heerspink, H. J. L., et al. (2023). Renal outcomes with incretin-based therapies. The Lancet Diabetes & Endocrinology, 11(4), 273–285.
Holst, J. J., & Rosenkilde, M. M. (2020). GIP as a therapeutic target in diabetes and obesity: Insight from incretin co-agonists. Journal of Clinical Endocrinology & Metabolism, 105(8), e2710–e2716. https://doi.org/10.1210/clinem/dgaa272
Hotamisligil, G. S. (2017). Inflammation, metaflammation and immunometabolic disorders. Nature, 542(7640), 177–185. https://doi.org/10.1038/nature21363
Jastreboff, A. M., Aronne, L. J., Ahmad, N. N., Wharton, S., Connery, L., Alves, B., Kiyosue, A., Zhang, S., Liu, B., & Stefanski, A. (2022). Tirzepatide once weekly for the treatment of obesity. New England Journal of Medicine, 387(3), 205–216. https://doi.org/10.1056/NEJMoa2206038
Kahn, S. E., Cooper, M. E., & Del Prato, S. (2014). Pathophysiology and treatment of type 2 diabetes: Perspectives on the past, present, and future. The Lancet, 383(9922), 1068–1083. https://doi.org/10.1016/S0140-6736(13)62154-6
Kapitza, C., et al. (2023). Efficacy and safety of tirzepatide: A systematic review and meta-analysis. Diabetes, Obesity and Metabolism, 25(5), 1231–1243.
Kelley, D. E., & Mandarino, L. J. (2000). Fuel selection in human skeletal muscle in insulin resistance. Diabetes, 49(5), 677–683. https://doi.org/10.2337/diabetes.49.5.677
Ludvik, B., Giorgino, F., Jódar, E., Frias, J. P., Fernández Landó, L., Brown, K., Bray, R., Rodríguez, Á., & SURPASS-3 Investigators. (2021). Once-weekly tirzepatide versus insulin degludec in type 2 diabetes. The Lancet, 398(10300), 583–598. https://doi.org/10.1016/S0140-6736(21)01443-5
Marso, S. P., Daniels, G. H., Brown-Frandsen, K., Kristensen, P., Mann, J. F. E., Nauck, M. A., Nissen, S. E., Pocock, S., Poulter, N. R., Ravn, L. S., Steinberg, W. M., Stockner, M., Zinman, B., Bergenstal, R. M., & LEADER Steering Committee. (2016). Liraglutide and cardiovascular outcomes in type 2 diabetes. The New England Journal of Medicine, 375(4), 311–322. https://doi.org/10.1056/NEJMoa1603827
Mensah, G. A., Roth, G. A., & Fuster, V. (2017). The global burden of cardiovascular diseases and risk factors. Journal of the American College of Cardiology, 70(20), 2529–2532. https://doi.org/10.1016/j.jacc.2017.09.009
Müller, T. D., Finan, B., Bloom, S. R., D’Alessio, D., Drucker, D. J., Flatt, P. R., Fritsche, A., Gribble, F., Grill, H. J., Habener, J. F., Holst, J. J., Langhans, W., Meier, J. J., Nauck, M. A., Perez-Tilve, D., Pocai, A., Reimann, F., Sandoval, D. A., Schwartz, T. W., … Tschöp, M. H. (2019). Glucagon-like peptide 1 (GLP-1). Molecular Metabolism, 30, 72–130. https://doi.org/10.1016/j.molmet.2019.09.010
Nauck, M. A., Quast, D. R., Wefers, J., & Meier, J. J. (2021). GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Molecular Metabolism, 46, 101102. https://doi.org/10.1016/j.molmet.2020.101102
Popay, J., Roberts, H., Sowden, A., Petticrew, M., Arai, L., Rodgers, M., Britten, N., Roen, K., & Duffy, S. (2006). Guidance on the conduct of narrative synthesis in systematic reviews. ESRC Methods Programme.
Samms, R. J., Coghlan, M. P., & Sloop, K. W. (2021). How may GIP enhance the therapeutic efficacy of GLP-1? Trends in Endocrinology & Metabolism, 32(7), 410–421. https://doi.org/10.1016/j.tem.2021.03.002
Santos, J. L., Boutin, P., Verdich, C., Holst, J. J., Larsen, L. H., Toubro, S., Astrup, A., & Froguel, P. (2023). Genetic variation in incretin receptors and response to GLP-1-based therapies. Diabetologia, 66(2), 201–213. https://doi.org/10.1007/s00125-022-05840-5
Sanyal, A. J., et al. (2024). Effects of dual incretin agonists on liver fat and inflammation. Hepatology, 79(1), 120–134.
Sattar, N., McGuire, D. K., & Packer, M. (2022). Cardiovascular safety and benefits of tirzepatide in type 2 diabetes. Nature Reviews Cardiology, 19, 665–666. https://doi.org/10.1038/s41569-022-00734-0
Smits, M. M., & Van Raalte, D. H. (2021). Safety of semaglutide. Frontiers in Endocrinology, 12, 645563. https://doi.org/10.3389/fendo.2021.645563
Thomas, J., & Harden, A. (2008). Methods for the thematic synthesis of qualitative research in systematic reviews. BMC Medical Research Methodology, 8, 45. https://doi.org/10.1186/1471-2288-8-45
Wadden, T. A., Bailey, T. S., Billings, L. K., Davies, M., Frias, J. P., Koroleva, A., Lingvay, I., O’Neil, P. M., Rubino, D. M., Skovgaard, D., Wallenstein, S., & Garvey, W. T. (2020). Effect of subcutaneous semaglutide on body weight and quality of life. JAMA, 323(12), 1173–1184. https://doi.org/10.1001/jama.2020.0928
Wagner, E. H., Austin, B. T., Davis, C., Hindmarsh, M., Schaefer, J., & Bonomi, A. (2001). Improving chronic illness care: Translating evidence into action. Health Affairs, 20(6), 64–78. https://doi.org/10.1377/hlthaff.20.6.64
Wilding, J. P. H., et al. (2022). Weight regain after discontinuation of GLP-1 therapy. Diabetes, Obesity and Metabolism, 24(8), 1553–1562.
Wilding, J. P. H., Batterham, R. L., Calanna, S., Davies, M., Van Gaal, L. F., Lingvay, I., McGowan, B. M., Rosenstock, J., Tran, M. T. D., Wadden, T. A., Wharton, S., Yokote, K., Zeuthen, N., Kushner, R. F., & STEP 1 Study Group. (2022). Sustained weight loss with semaglutide and implications for long-term obesity management. Diabetes, Obesity and Metabolism, 24(8), 1553–1562. https://doi.org/10.1111/dom.14649